Calprotectin Immunoturbidimetric (IT) Assay
FDA-cleared and IVDD for the quantitative determination of calprotectin in human stool, in vitro diagnostic use as an aid in the diagnosis of inflammatory bowel disease (IBD), specifically Crohn’s disease (CD) and ulcerative colitis (UC), and as an aid in the differentiation of IBD from irritable bowel syndrome (IBS) in conjunction with other clinical and laboratory findings.
ALPCO’s Calprotectin IT Assay combines the clinical sensitivity and specificity clinicians expect with the efficiency and repeatability today’s laboratories require.
Take Control of Your Calprotectin Testing
See for yourself why labs are switching to ALPCO’s new FDA-cleared Calprotectin Immunoturbidimetric Assay
One free kit per laboratory. Void where prohibited.


Clinically Accurate
- Clinical sensitivity of 90.5% - few false negatives
- Clinical specificity of 93.4% - few false positives


Reliable
- AMR of 11 -10,000 µg/g* - broad range of reportable quantitative results
- Inter-lot CV of only 4.1% - repeatable performance


Fast
- Testing of >400 samples / hour** - high throughput
- STAT results in 10 minutes** - fast turn-around


User Friendly
- Compatible with standard clinical chemistry analyzers - minimal training and hands-on time
- Universal stool extraction device - single preparation with no manual weighing


Cost Effective
- Only 2 non-reportables per day - minimal cost for low and high volume testing
- Cost savings vs. send-out with revenue potential and CAGR of 6%
*With automatic 1:10 dilution
**Based on the Beckman AU680 analyzer
Your Fecal Calprotectin Testing, Elevated.
No matter how you are currently running your fecal calprotectin testing, ALPCO's new Calprotectin Immunoturbidimetric Assay will take your testing to the next level.
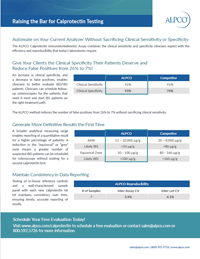
Sending Out for Calprotectin Testing? Bring Testing In-House
- Utilize Your Existing Clinical Chemistry Analyzer
- Improve Turn-Around Time
- Decrease Cost and Increase Profitability
Testing Fecal Calprotectin on a CLIA System?
- Clinical Chemistry Analyzer for Calprotectin
- Free Space on Your CLIA System for Menu Expansion
- Increase Throughput and Walk-Away Time for Calprotectin Testing
Using Another Immunoturbidimetric Assay?
- Reduce False Positives
- Generate More Definitive Results the First Time
- Maintain Consistency in Data Reporting
Running a Traditional Calprotectin ELISA?
- Utilize an Existing Clinical Chemistry Analyzer
- Increase Laboratory Efficiency
- Improve Turn-Around Time
Products for Gastroenterology
-
 ALEX³ Allergy Xplorer Kit (20 tests)
ALEX³ Allergy Xplorer Kit (20 tests)Catalog #: 87-03-2001-02
Sample Types: Citrate Plasma, Heparin Plasma, Serum
Sample Size(s): 100 µL
-
 ALEX³ Allergy Xplorer Kit (20 tests)
ALEX³ Allergy Xplorer Kit (20 tests)Catalog #: 87-03-2001-01
Sample Types: Citrate Plasma, Heparin Plasma, Serum
Sample Size(s): 100 µL
-
 ALEX³ Allergy Xplorer Kit (50 tests)
ALEX³ Allergy Xplorer Kit (50 tests)Catalog #: 87-03-5001-02
Sample Types: Citrate Plasma, Heparin Plasma, Serum
Sample Size(s): 200 µL
-
 ALEX³ Allergy Xplorer Kit (50 tests)
ALEX³ Allergy Xplorer Kit (50 tests)Catalog #: 87-03-5001-01
Sample Types: Citrate Plasma, Heparin Plasma, Serum
Sample Size(s): 200 µL
-
 FOX Food Xplorer Kit (50 tests)
FOX Food Xplorer Kit (50 tests)Catalog #: 87-80-5001-02
Sample Types: Plasma, Serum
Sample Size(s): 100 µL
-
 ImageXplorer Bundle Canada
ImageXplorer Bundle CanadaCatalog #: 87-IXBUNDLE-01
-
 Quality Xplorer ALEX² sIgE Allergy Control
Quality Xplorer ALEX² sIgE Allergy ControlCatalog #: 87-31-0800-02