January 6, 2015
Vitamin D Metabolism and Function
Understanding Vitamin D Metabolism & Function
Vitamin D is a steroid hormone involved in the intestinal absorption of calcium and the regulation of calcium homeostasis, bone differentiation and immune response. The term "vitamin D" refers to several different forms of this vitamin. There are two major types of vitamin D:- Vitamin D2 (ergocalciferol) – which is synthesized by plants and is not produced by the human body.
- Vitamin D3 (cholecalciferol) – which is made in large quantities in the skin when sunlight strikes bare skin. It can also be ingested from animal sources.
Video produced by Animated Technologies for Abbott Labs
The storage type of vitamin D, namely 25- hydroxy vitamin D, is formed in the liver. The hormone 1,25-dihydroxy vitamin D (D hormone) is formed in a second hydroxylation step in the kidney. The responsible enzyme, the kidney 1 α-hydroxylase, is subjected to a rigid control through hormones (especially parathyroid hormone) and its activity is influenced by the serum concentrations of calcium and phosphate.
Forms of Vitamin D
25 Hydroxy
25-OH Vitamin D plays a key role in the maintenance of mineral balance and skeletal integrity. Long associated only with its role in bone formation; Vitamin D is actually active throughout the human body, influencing immune system responses and cell defenses. 25-Hydroxyvitamin D (25OHD) is the major circulating form of vitamin D and the precursor of the active form (1,25-dihydroxyvitamin D). Because of its long half-life, 25OHD measurements are useful for assessing vitamin D status.Vitamin D occurs in 2 forms: vitamin D3 (cholecalciferol) and vitamin D2 (ergocalciferol). Most current methods do not differentiate between the 2 forms: only total 25OHD concentrations are reported. The reference range for total 25OHD (20-100 ng/mL) is based on 25OHD correlation with physiological parameters that include parathyroid hormone concentration and calcium absorption.4-8 The range is not based on the distribution of levels in an apparently healthy population. 25OHD2– and 25OHD3– specific reference ranges are not available. Total Vitamin D (25-hydroxy) Interpretation Ranges1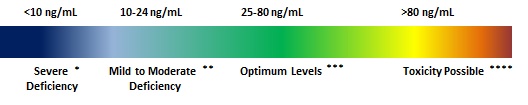 These reference ranges represent clinical decision values that apply to males and females of all ages, rather than population-based reference values.
* Could be associated with osteomalacia or rickets
** May be associated with increased risk of osteoporosis or secondary hyperparathyroidism
*** Optimum levels in the normal population
**** 80 ng/mL is the lowest reported level associated with toxicity in patients without primary hyperparathyroidism who have normal renal function.
These reference ranges represent clinical decision values that apply to males and females of all ages, rather than population-based reference values.
* Could be associated with osteomalacia or rickets
** May be associated with increased risk of osteoporosis or secondary hyperparathyroidism
*** Optimum levels in the normal population
**** 80 ng/mL is the lowest reported level associated with toxicity in patients without primary hyperparathyroidism who have normal renal function.
1,25 Dihydroxy
Of all the steroid hormones, 1,25(OH)2D represented the most difficult challenge to the analytical biochemist with respect to quantitation. 1,25(OH)2D circulates at pmol concentrations (too low for direct UV or MS quantitation), is highly lipophilic and its precursor, 25(OH)D, circulates at nmol levels.2 1,25-Dihydroxy vitamin D (aka Calcitiol) is the most potent vitamin D metabolite. It stimulates calcium absorption in the intestine and its production is tightly regulated through concentrations of serum calcium, phosphorus, and parathyroid hormone. Calcitriol is produced in the cells of the proximal tubule of the nephron in the kidneys by the action of 25-hydroxyvitamin D3 1-alpha-hydroxylase, a mitochondrial oxygenase and an enzyme which catalyzes the hydroxylation of 25-hydroxycholecalciferol (calcifediol). The activity of the enzyme is stimulated by PTH. The reaction is an important control point in Ca2+ homeostasis.3 Calcitriol increases blood calcium levels ( [Ca2+] ) by promoting absorption of dietary calcium from the gastrointestinal tract and increasing renal tubular reabsorption of calcium thus reducing the loss of calcium in the urine. Calcitriol also stimulates release of calcium from bone by its action on the specific type of bone cells referred to as osteoblasts, causing them to release RANKL, which in turn activates osteoclasts.4 Calcitriol acts in concert with parathyroid hormone (PTH) in all three of these roles. For instance, PTH also stimulates osteoclasts. However, the main effect of PTH is to increase the rate at which the kidneys excrete inorganic phosphate (Pi), the counterion of Ca2+. The resulting decrease in serum phosphate causes Ca5(PO4)3OH to dissolve out of bone thus increasing serum calcium. PTH also stimulates the production of calcitriol.3 Various research has shown 1,25 levels as an indication for:- Disturbances of kidney functions:
- Chronic kidney failure
- Haemodialysis following kidney transplants
- Renal osteopathy
- Osteomalacia from various types of vitamin D metabolism disturbances
- Monitoring of therapy with active vitamin D metabolites
- Idiopathic hypercalciuria
- Hypercalcaemia